Dodge Journey: Description, Operation
DESCRIPTION
There are two different batteries available for this vehicle. Vehicles equipped with a diesel engine utilize a spiral cell battery with recombination technology. This is a maintenance-free battery that is capable of delivering more power than a conventional battery. This additional power is required by a diesel engine during cold cranking. Vehicles equipped with a gasoline engine utilize a conventional battery. Refer to the following information for detailed differences and descriptions of these two batteries.
SPIRAL CELL BATTERY - DIESEL ENGINE
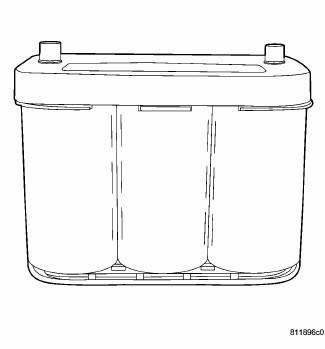
Fig. 7: Spiral Plate Battery - Typical
WARNING: Never exceed 14.4 volts when charging a spiral cell battery. Personal injury and/or battery damage may result.
By tightly winding layers of spiral grids and acid-permeated vitreous separators into cells, the battery has more power and service life than conventional batteries of the same size. The spiral cell battery is completely, permanently sealed. Through gas recombination, hydrogen and oxygen within the battery are captured during normal charging and reunited to form the water within the electrolyte, eliminating the need to add distilled water. Therefore, these batteries have non-removable battery vent caps.
The acid inside a spiral cell battery is bound within the vitreous separators, ending the threat of acid leaks. This feature allows the battery to be installed in any position anywhere in the vehicle.
Spiral cell technology is the process by which the cells holding the active material in the battery are wound tightly in coils instead of hanging flat, like conventional batteries. This design has a lower internal resistance and also increases the active material surface area.
Due to the maintanance-free design, distilled water cannot be added to this battery. Therefore, if more than 14.4 volts are used during the spiral cell battery charging process, water vapor can be exhausted through the pressure-sensitive battery vents and lost for good. This can permanently damage the spiral cell battery.
CONVENTIONAL BATTERY - GASOLINE ENGINE
Low-maintenance conventional batteries are used on vehicles equipped with a gasoline engine, these batteries have non-removable battery cell caps. Under normal service, the composition of this battery reduces gassing and water loss at normal charge rates.
Conventional batteries are made up of six individual cells that are connected in series. Each cell contains positive charged cell groups made of lead oxide, and negatively charged cell groups made of sponge lead. The cells are submerged in a sulfuric acid and water solution called electrolyte.
The battery is used to store electrical energy potential in a chemical form. When an electrical load is applied to the battery terminals, an electrochemical reaction occurs within the battery. This reaction causes the battery to discharge electrical current.
OPERATION
The battery is designed to store electrical energy in a chemical form. When an electrical load is applied to the terminals of the battery, an electrochemical reaction occurs. This reaction causes the battery to discharge electrical current from its terminals. As the battery discharges, a gradual chemical change takes place within each cell. The sulfuric acid in the electrolyte combines with the plate materials, causing both plates to slowly change to lead sulfate. At the same time, oxygen from the positive plate material combines with hydrogen from the sulfuric acid, causing the electrolyte to become mainly water. The chemical changes within the battery are caused by the movement of excess or free electrons between the positive and negative plate groups. This movement of electrons produces a flow of electrical current through the load device attached to the battery terminals.
As the plate materials become more similar chemically, and the electrolyte becomes less acid, the voltage potential of each cell is reduced. However, by charging the battery with a voltage higher than that of the battery itself, the battery discharging process is reversed. Charging the battery gradually changes the sulfated lead plates back into sponge lead and lead dioxide, and the water back into sulfuric acid. This action restores the difference in the electron charges deposited on the plates, and the voltage potential of the battery cells. For a battery to remain useful, it must be able to produce high-amperage current over an extended period. A battery must also be able to accept a charge, so that its voltage potential may be restored.
The battery is vented to release excess hydrogen gas that is created when the battery is being charged or discharged. However, even with these vents, hydrogen gas can collect in or around the battery. If hydrogen gas is exposed to flame or sparks, it may ignite. If the electrolyte level is low, the battery may arc internally and explode. If the battery is equipped with removable cell caps, add distilled water whenever the electrolyte level is below the top of the plates. If the battery cell caps cannot be removed, the battery must be replaced if the electrolyte level becomes low.
 Battery
Battery
...
 Diagnosis and testing
Diagnosis and testing
BATTERY
The battery must be completely charged and the terminals should be properly
cleaned and inspected before
diagnostic procedures are performed. See Cleaning for the proper battery
cleaning ...
See also:
DEALER SERVICE
Your authorized dealer has the qualified service personnel,
special tools, and equipment to perform all service
operations in an expert manner. Service Manuals are
available which include detailed ...
Operation
FIXED DISPLACEMENT A/C COMPRESSOR
The Denso 10SR15 and 10SR17 fixed displacement A/C compressors are driven by
the engine through an
electric clutch, drive pulley and belt arrangement. These A/C c ...
Switch, ignition
DESCRIPTION
This vehicle is equipped with a Wireless Ignition Node (WIN). The WIN, along
with the FOB with Integrated
Key (FOBIK) are the primary components of the keyless ignition system. The onl ...
